See the Thinking
Compliance work can feel abstract until you see it applied. These examples show the process, the thinking, and the kind of clarity a founder walks away with.
PDP diagnostic: omega-3 supplement
A product page that felt great at launch can become a problem as a brand starts pushing into paid traffic, retail, or new sales channels. What reads as natural and persuasive to a founder (outcome-led, emotional, benefits-first) is usually the exact wording that regulators flag.
A PDP Diagnostic pinpoints that risk before it turns into a CAP/ASA headache.
In this example, I’m showing the deliverables for a fictional omega-3 brand making heart-health claims. I review the full product page and assess every section against UK CAP/ASA guidance and EC Regulation 1924/2006. That review produces two outputs:
1. A colour-coded wireframe of the full page.
Each content block is marked high, medium, or low risk, so the founder immediately sees where the problems sit and which areas are already in safe territory.
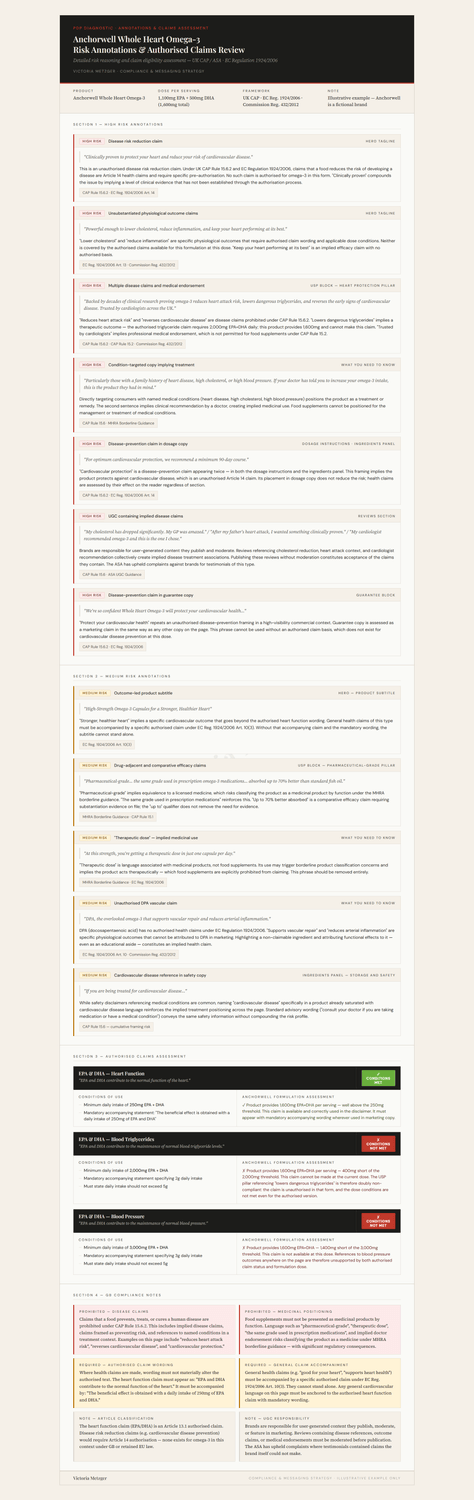
2. A detailed annotations document.
Every high- and medium-risk issue is broken down clearly: what the issue is, the specific rule it triggers, and which authorised claim wording the brand can legally use at its current formulation dose.
In this example, the page includes seven high-risk issues, from disease claims to implied medical endorsement to condition-led messaging. And although the brand wants to use three EPA/DHA heart-related claims, the assessment shows that only one is actually permitted at their dose.
The founder walks away with a clean, prioritised plan: what needs changing, what’s fine as is, and what’s possible if they want to reformulate or reposition.
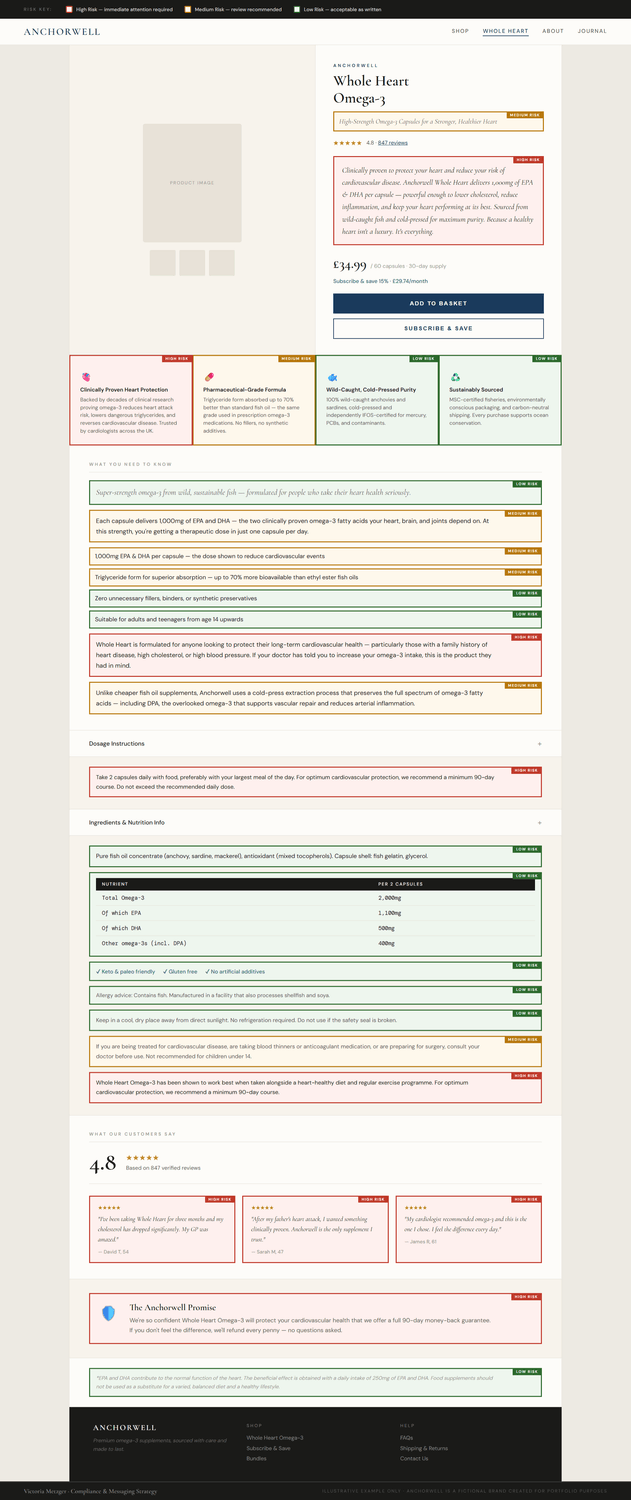
A colour-coded wireframe of the full product page.
Wondering where your product page sits? Book a focused assessment that shows you exactly where your claims are exposed and what needs addressing before you scale.
Pre-launch messaging review: caffeine support supplement
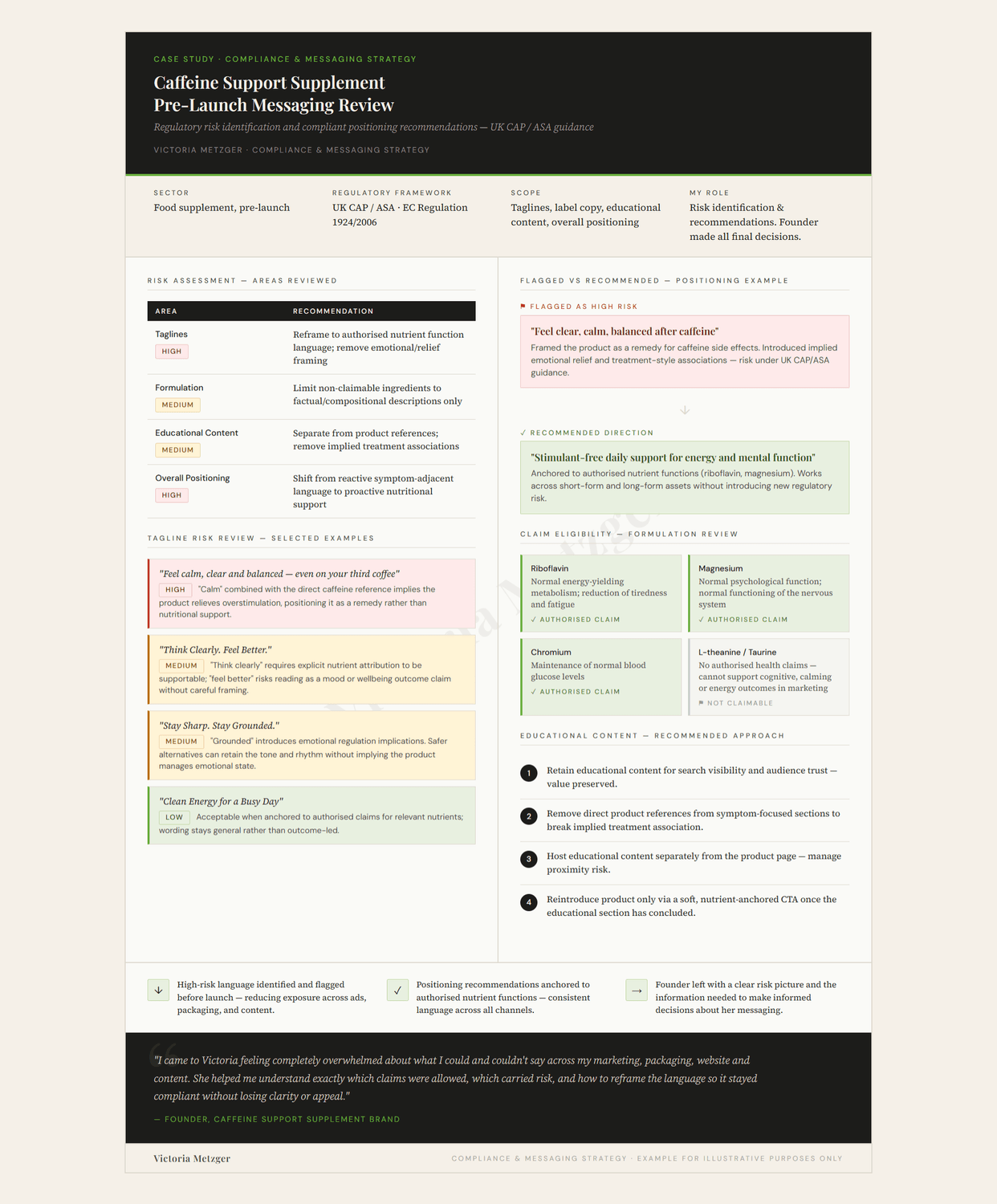
This infographic illustrates the risk assessment framework applied during the review, showing the areas examined, how individual taglines were assessed, and the reasoning behind the positioning recommendations provided.
A supplement brand targeting consumers who enjoy caffeine but are sensitive to its effects. The founder had a strong concept and clear brand voice. The difficulty was in how that idea showed up in the language: early messaging framed the product too closely as a solution to caffeine side effects, creating implied symptom-relief risk under UK CAP/ASA guidance.
The work involved reviewing proposed taglines, educational content, and formulation-level claim eligibility, then identifying how the positioning could be supported by authorised UK health claims without losing the brand's original feel.
High-risk language was flagged with recommended alternatives before launch. Educational content was identified as retainable for SEO value, with separation from product references recommended to remove implied treatment associations.
The founder left with a clear picture of where the risks sat and what her options were, giving her the information she needed to make confident decisions about her messaging across ads, packaging, and content.
MENU
The information and services provided by VJM Writing Services are for educational and marketing purposes only and do not constitute legal or regulatory advice. While we create content designed to align with regulatory frameworks (ASA, EFSA, FDA, FTC, etc.), compliance involves many factors beyond marketing copy, including formulations, labelling, and internal processes. We cannot guarantee product approvals or prevent regulatory action, and we recommend consulting a qualified legal or regulatory professional for full compliance support.
© VJM Writing Services 2026

